Tutorial covering the formation and reactivity of sulfonate esters, such as tosylates and mesylates.
Introduction
Alkyl halides undergo elimination reactions and nucleophilic substitution reactions, in which a carbon-halogen bond is broken and a halide ion is released. These reactions are favourable for alkyl halides, as halogens are (usually) good leaving groups (halide ions are weak bases).
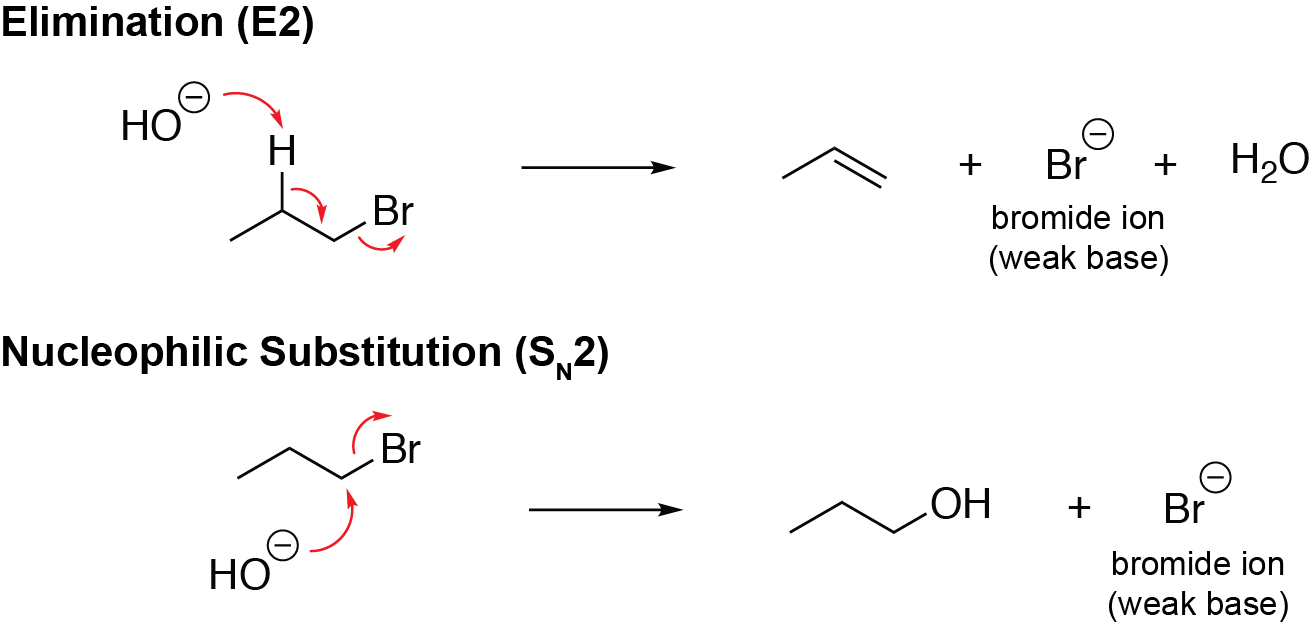
Alcohols can also react through elimination and nucleophilic substitution mechanisms, in which the carbon-oxygen bond is broken and hydroxide is the leaving group. However, these reactions are not very favourable, as the hydroxyl group is not a very good leaving group (hydroxide is a strong base).
Hydroxyl groups can be made into better leaving groups by using a strong acid. For example, in the E1 alcohol dehydration mechanism, the alcohol hydroxyl group is protonated by the strong acid, the carbon-oxygen bond breaks, and a water molecule is released. Protonating a hydroxyl group makes it into a far better leaving group (water is a weak base).

Another way that alcohols can be made into good leaving groups is by converting them into sulfonate esters.
Sulfonate Esters
Similar to the relationship between (carboxylic acid) esters and carboxylic acids, sulfonic esters are derivatives of sulfonic acids. Sulfonate esters contain a sulfur with two double bonds to oxygens, a single bond to an R group, and a single bond to an OR group (where the R groups are usually alkyl or aryl groups). Three sulfonate esters are commonly used in organic chemistry: the p-toluenesulfonate ester (or tosylate), the methanesulfonate ester (or mesylate), and the trifluoromethanesulfonate ester (or triflate).

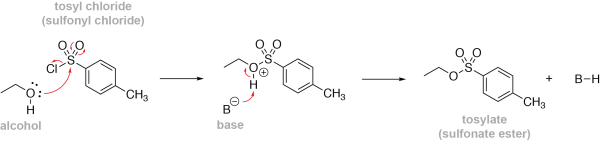
Alcohols can be converted into sulfonate esters by treating them with a sulfonyl chloride and a base. For example, to form a tosylate, p-toluenesulfonyl chloride (or tosyl chloride) is used. Sulfonyl chlorides are very good electrophiles, analogous to acyl chlorides. The hydroxyl group of an alcohol nucleophilically attacks the sulfur of a sulfonyl chloride, displacing the chloride, and forming a sulfonate ester. A base, such as pyridine, can then gather up the acidic proton that is released, preventing the production of HCl as a by-product. Here’s a mechanism showing the formation of the tosylate from a simple alcohol:
By converting an alcohol into a sulfonate ester, its reactivity becomes like that of an alkyl halide. For this reason, sulfonate esters are sometimes called pseudohalides. Activated as a sulfonate ester, alcohols undergo elimination and nucleophilic substitution reactions much more easily. These examples show bimolecular elimination and nucleophilic substitution reactions for a tosylate and a mesylate:

Why are sulfonate esters such good leaving groups? Once the carbon-oxygen bond is broken in these mechanisms, a negatively-charged sulfonate is produced. The negative charge is delocalized between the three oxygens on the sulfur (that is, there is resonance stabilization), and so this is a very weak base.
Summary
It is not favourable for alcohols to undergo elimination and nucleophilic substitution reactions, because hydroxyl groups are poor leaving groups. However, alcohols can be made to undergo these reactions by converting them into sulfonate esters, which are much better leaving groups. Alcohols are converted into sulfonate esters by treating them with a sulfonyl chloride and base. The most commonly used sulfonate esters are tosylates (p-toluenesulfonate esters), mesylates (methanesulfonate esters), and triflates (trifluoromethanesulfonate esters).