Tutorial on the alkene hydrohalogenation reaction, which converts alkenes to alkyl halides using hydrogen halides.
Reaction
The alkene hydrohalogenation reaction is an electrophilic addition reaction which converts alkenes into alkyl halides. The alkene reacts with a hydrogen halide (HX, where X is Cl, Br, or I), breaking the alkene π bond, adding a hydrogen to one alkene carbon, and adding a halogen atom (Cl, Br, or I) to the other.
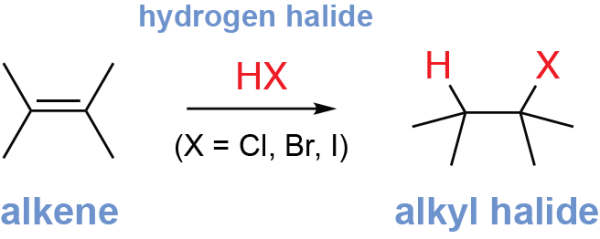
The position where the halogen is added can be predicted by Markovnikov’s rule. This rule states that, in an electrophilic addition reaction, the nucleophile (here, the halogen) will be added to the more highly substituted alkene carbon (the alkene carbon that has more alkyl groups) to give the Markovnikov product.

Mechanism
The mechanism of alkene hydrohalogenation is very similar to other electrophilic addition reactions, especially the alkene hydration reaction. Let’s go through the mechanism of this reaction, where an alkene reacts with hydrogen bromide (HBr) to produce an alkyl halide:
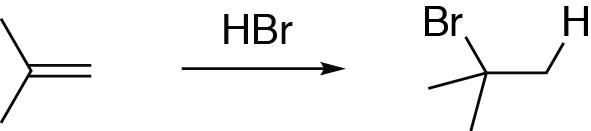
Like other electrophilic addition reactions, this mechanism begins with the nucleophilic attack of the alkene π electrons (i.e., the double bond) onto an electrophile. Here, the electrophile is the hydrogen atom of HBr, which carries a partial positive charge. Draw an arrow from the alkene double bond to the hydrogen atom of hydrogen bromide. This leads to the cleavage of the H-Br bond, as shown with an arrow going from the H-Br bond to the bromine. This results in a negatively charged bromide ion.
The hydrogen from HBr is added to one of the alkene carbons, while the other alkene carbon gets a positive charge. The position that becomes positively charged is determined based on carbocation stability. If the carbon on the right is protonated, a tertiary carbocation is formed (top arrow), and if the carbon of the left is protonated, a primary carbocation is formed (bottom arrow):
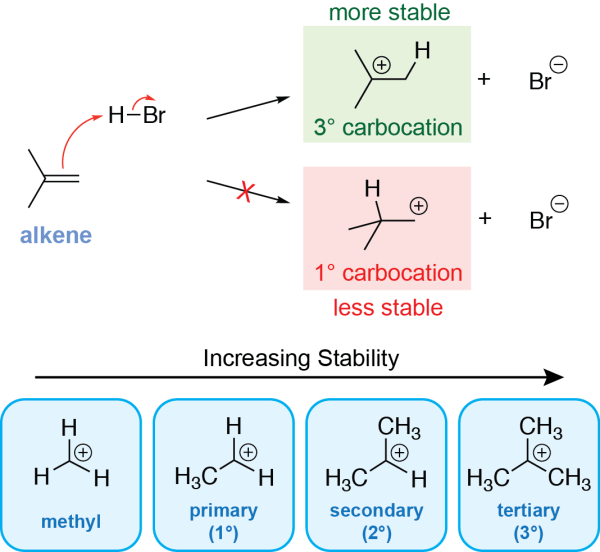
As carbocations with more substituents are more stable, the top reaction arrow is much more favourable, and so the tertiary carbocation is the major product of the first step.
Now that the carbocation formed, the second step is the nucleophilic attack of the bromide ion onto the electrophilic carbocation, forming the alkyl halide product. This is shown by drawing an arrow from the bromide ion to the carbocation carbon:
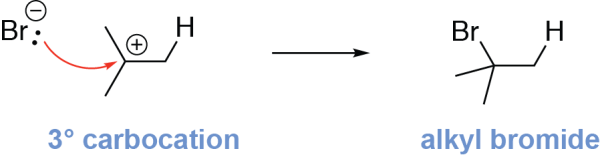
Markovnikov’s rule, which predicts that the nucleophile in an electrophilic addition reaction will add to the more highly substituted alkene carbon, is based on carbocation stability. In these reactions, formation of the more substituted (and thus more stable) carbocation intermediate is preferred. As the more substituted carbocation is preferentially formed, the nucleophile will add to this carbocation, and so the nucleophile is added to the more substituted carbon.
In the alkene hydrohalogenation reaction, two different products are possible, depending on which alkene carbon the halogen atom is added to. These two products are regioisomers, which are structural isomers that differ based on the position of a substituent or functional group (in this case, the halogen atom). As this reaction follows Markovnikov’s rule, one regioisomer is usually the major product – thus, this reaction is said to be regioselective, and the Markovnikov product regioisomer is preferentially formed. For other alkenes, in which both alkene carbons have the same number of substituents, this reaction may be less regioselective, and can give a mixture of products.
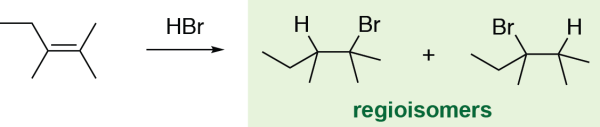
Note that some alkene reactions, such as the hydroboration-oxidation reaction, preferentially form the anti-Markovnikov product, in which the nucleophile ultimately ends up on the less substituted alkene carbon.
Summary
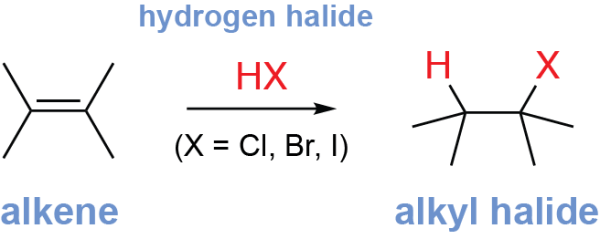
The alkene hydrohalogenation reaction converts an alkene into an alkyl halide, using a hydrogen halide. This is an electrophilic addition reaction where the alkene nucleophilically attacks an electrophile, a carbocation is formed, and a second nucleophile attacks the carbocation. This reaction follows the Markovnikov’s rule, as the carbocation is preferentially formed on the more highly substituted alkene carbon. This is thus a regioselective reaction, as one regioisomer is usually the major product (the Markovnikov product).
Related
Alkene Hydration Reaction – a related electrophilic addition reaction, which converts alkenes into alcohols.