Next: Formal Charges and Mechanisms (Part 2)
Previous: Formal Charges and Mechanisms (Part 1)
This tutorial explains how the mechanisms of simple chemical reactions are represented by curved arrows.
Drawing Curved Arrows for Reaction Mechanisms
The previous tutorial demonstrated how a single curved arrow can represent the movement of electrons. However, the mechanisms of actual organic reactions are more complex, and usually must be represented by more than one curved arrow.
In most organic mechanisms, when a bond is made, another bond must be broken. The number of bonds an atom can form is limited by its number of valence electrons, which are either held as lone pairs, or are shared in bonds. If a new bond is formed involving an atom, and there is no change in the number of lone pairs on that atom, another bond will usually be broken.
The following examples build on the simple mechanisms covered in part 1. They were chosen because they highlight the different ways that curved arrows can be drawn outlined in part 1 (from lone pair to atom, from lone pair to adjacent bond, from bond to adjacent atom, from bond to adjacent bond, and from bond to unattached atom). Don’t worry about the specific reactions for now, it’s more important to see how the curved arrows show the formation and cleavage of bonds
Note the conservation of charge: the net charge of the structures before the reaction arrow matches the net charge of the structures after the reaction arrow. Also note how the formal charge of an atom at (or near) the start of the curved arrows increases, and the formal charge of the atom at the end of the curved arrows decreases.
Arrow from lone pair to atom
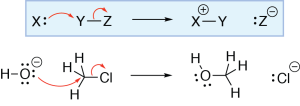
In this reaction, a hydroxide ion is reacting with an alkyl halide. One of the lone pairs on the hydroxide ion oxygen atom is making a σ bond with the carbon of the alkyl halide, so we draw a curved arrow from an oxygen lone pair to the carbon. The oxygen has lost a lone pair and gained a bond, so its formal charge increases by one, and it becomes neutral. Check the previous tutorial, Formal Charges and Mechanisms (Part 1), for an explanation of how formal charges can change during a reaction mechanism.
Carbon (like all other second row elements) can have a maximum of four bonds. For the oxygen-carbon bond to form, another bond on the carbon must be broken. In this reaction, the carbon-chlorine σ bond breaks, and the electrons from that bond become a lone pair on chlorine. This is shown by drawing a curved arrow from the carbon-chlorine bond to the chlorine. The chlorine atom has lost a bond and gained a lone pair, so its formal charge decreases by one and it becomes negatively charged. Note that the formal charge of the carbon does not change; although it has gained a bond, it has also lost a bond.
Arrow from lone pair to adjacent bond
In this reaction, the negatively charged oxygen is making a carbon-oxygen π bond. So, we draw a curved arrow from the oxygen lone pair to the carbon-oxygen σ bond. As the oxygen has lost a lone pair and gained a bond, its formal charge increases by one, and it becomes neutral.
As carbon can have a maximum of four bonds, another bond on the carbon must be broken. In this reaction, the carbon-chlorine σ bond is cleaved, and the bond electrons become a lone pair on the chlorine. This is shown by drawing a curved arrow from the carbon-chlorine bond to the chlorine, giving it a lone pair. The chlorine has gained a lone pair and lost a bond, so its formal charge decreases by one, and it becomes negatively charged. Again, the formal charge of the carbon stays the same, as there is no net change in its number of bonds and lone pairs.
Arrow from bond to adjacent atom
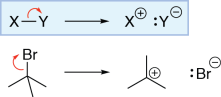
In this reaction, the carbon-bromine σ bond is broken, and the bond electrons become a lone pair on bromine. This is shown by drawing a curved arrow from the carbon-bromine bond to the bromine atom. The carbon has lost a bond, but has not gained a lone pair, so its formal charge increases by one and it becomes positively charged. The bromine has lost a bond and gained a lone pair, so its formal charge decreases by one and it becomes negatively charged.
Arrow from bond to adjacent bond
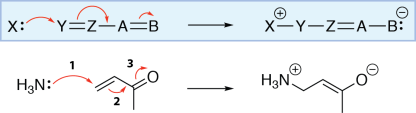
Here’s a more complicated mechanism, which shows the reaction of ammonia with an α,β-unsaturated ketone. For now, all we care about are the curved arrows, so don’t worry about the overall reaction.
This reaction starts with the ammonia nitrogen forming a σ bond with the carbon at the end of the chain. This is shown by drawing curved arrow 1 from the nitrogen lone pair to the carbon.
As carbon can only have a maximum of four bonds, another bond must be broken. Here, the carbon-carbon π bond is broken, and the π electrons move to make a new π bond in the adjacent position. We show this by drawing curved arrow 2 from the carbon-carbon π bond to the neighbouring carbon-carbon σ bond.
However, to make this new carbon-carbon π bond, another bond must be broken on the carbon double-bonded to the oxygen. In this reaction, the carbon-oxygen π bond breaks, and the electrons from the π bond become a lone pair on the oxygen. We show this by drawing curved arrow 3 from the carbon-oxygen π bond to the oxygen.
The nitrogen at the start of the reaction has lost a lone pair and gained a bond, so its formal charge increases by one. The oxygen at the end of the reaction has gained a lone pair and lost a bond, so its formal charge decreases by one. There is no net change in the number of bonds and lone pairs on the carbons in this molecule, so their formal charges stay the same.
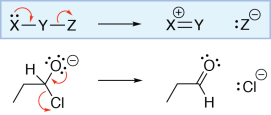
Arrow from bond to unattached atom
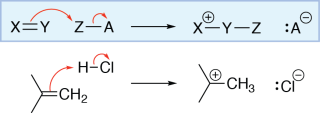
In this reaction, an alkene is reacting with hydrochloric acid (HCl). The electrons in the π bond of the alkene are being used to make a σ bond between one of the alkene carbons and the hydrogen of HCl. This can be shown by drawing a curved arrow from the carbon-carbon π bond to the hydrogen of HCl.
As hydrogen can only have one bond, the hydrogen-chlorine bond must be broken. To show this, we draw an arrow from the bond to the chlorine atom, giving it a lone pair. As the chlorine atom gains a lone pair and loses a bond, its formal charge decreases by one. Note that the other alkene carbon (which did not form a bond with the hydrogen) only has three bonds. It has lost a bond and has not gained a lone pair, so its formal charge increases by one.
Related Tutorials
The previous tutorials, Drawing Curved Arrows (Part 1) and Formal Charges and Mechanisms (Part 1), introduce the topics covered in this tutorial.
The next tutorial, Formal Charges and Mechanisms (Part 2), explains in more detail how the formal charges of carbon, nitrogen, and oxygen change in a reaction mechanism.