Next: Drawing Curved Arrows (Part 2)
Previous: Drawing Curved Arrows (Part 1)
This tutorial explains how the formal charges of atoms can change in a reaction mechanism, resulting from the movement of electrons.
Introduction
Curved arrows are used to represent the movement of electrons (in bonds and lone pairs) in a reaction mechanism. Polar reactions involve the movement of pairs of electrons, which can be represented using double-headed curved arrows.
The formal charge of an atom in a molecule is a value describing the electrical charge of that atom. Formal charge can be calculated based on the number of valence electrons, lone pairs, and bonds on an atom.
Curved Arrows and Formal Charges
The movement of electrons in a reaction mechanism can result in changes to the formal charges of the atoms involved. However, it would be tedious to calculate the formal charge of every atom in every step of the mechanism. Instead, changes to formal charge can be determined based on changes in the number of lone pairs and bonds on an atom. If the number of bonds and lone pairs on an atom does not change, the formal charge of that atom does not change.
The two electrons in a lone pair on an atom belong entirely to that atom. However, the two electrons in every σ and π bond are shared between the two bonded atoms. When evaluating formal charge, you can consider that one electron in each bond belongs to each atom.
There is a limited number of ways that bonds can be formed and broken in an organic reaction mechanism. The impact that electron movement has on formal charge usually falls into one of the following four situations; note how the changes in formal charge are related to the position of the curved arrow:
- If a lone pair on an atom is used to make a bond, the charge of that atom increases by one. The atom initially had a lone pair (two electrons), but those electrons are now shared in a bond (so it now has one electron). The atom has lost an electron, so its charge increases by one.
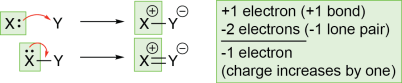
- If an atom gains a lone pair after a bond breaks, the charge of that atom decreases by one. The atom initially owned just one electron in the bond, but it has taken both electrons from the bond and made a lone pair. The atom has gained an electron, so its charge decreases by one.
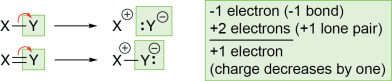
- If an atom does not gain a lone pair after a bond breaks, the charge on that atom increases by one. The atom initially owned one electron in the bond, but it lost possession of this electron when the bond broke. The atom has lost an electron, so its charge increases by one.
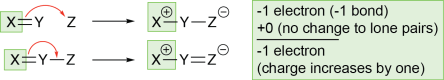
- If an atom gains a new bond without losing a lone pair, the charge of that atom decreases by one. The atom now owns an electron in the new bond without having given anything up. The atom has gained an electron, so its charge decreases by one.
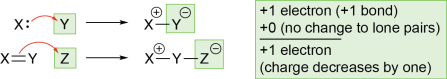
In these examples, you’ll see that an atom at (or near) the start of a double-headed curved arrow becomes more positively charged, and an atom at the end of the arrow becomes more negatively charged. This makes sense, as curved arrows show that negatively charged electrons are moving from the start of the arrow (making the start more positive) to the end of the arrow (making the end more negative).
Don’t worry about trying to categorize every curved arrow you see into one of these four categories. It’s more important to see how changes in the numbers of bonds and lone pairs result in changes to formal charge.
Although the above examples demonstrate how a single curved arrow affects formal charge, many organic reaction mechanisms involve more than one curved arrow. Often, the effect that one arrow has on formal charge will be compensated for by another arrow. Consider the following example:
![]()
While atom Y is getting a new bond with X, the bond between Y and Z is being broken at the same time. The net result is that the formal charge of Y does not change. As mentioned above, if the number of bonds and lone pairs on an atom remains the same, the formal charge of that atom will also stay the same.
Summary
The formal charge of an atom is determined by its number of valence electrons, lone pairs and bonds. If the number of lone pairs and bonds on an atom change in a reaction mechanism, the formal change of that atom will also change. Conversely, if the net result of two (or more) curved arrows in a reaction mechanism is that there is no change in the number of lone pairs and bonds on an atom, the formal charge of that atom will not change.
An atom can be considered to own two electrons from a lone pair, and one electron from a bond. By calculating the change in the number of electrons on an atom, based on changes in the number of lone pairs and bonds, the change to its formal charge can be determined.
Related Tutorials
The previous tutorial, Drawing Curved Arrows (Part 1), explained how curved arrows are used to show the movement of electrons in organic reaction mechanisms. The next tutorial, Drawing Curved Arrows (Part 2), gives examples showing the use of curved arrows to represent simple reaction mechanisms.
Formal Charges and Mechanisms (Part 2) further explains how formal charges can change in a reaction mechanism, focusing on carbon, nitrogen, and oxygen.