Next: Nucleophiles and Electrophiles
Previous: Formal Charges and Mechanisms (Part 2)
Introduction
The previous tutorial introduced the structures of some common charged carbon, nitrogen, and oxygen species encountered in reaction mechanisms. Some of these species are Brønsted acids and bases, as well as Lewis acids and bases.
Briefly, Brønsted acids are species that donate protons, and Brønsted bases are species that accept protons. Lewis acids are species that accept a pair of electrons, and Lewis bases are species that donate a pair of electrons.
In other words, a Lewis base must have an electron pair that it is able to donate, and a Lewis acid must be able to accept an electron pair. Certain functional groups are good Lewis bases, and others are good Lewis acids.
There is overlap between the Brønsted and Lewis definitions of acids and bases. How does a Brønsted base accept a proton? It makes a bond to the proton by donating a pair of electrons (i.e., it acts as a Lewis base). How does a Brønsted acid donate a proton? The acidic proton accepts a pair of electrons (i.e., it acts as a Lewis acid). Brønsted acids/bases can be considered a subset of Lewis acids/bases.
Many organic reactions start as interactions between Lewis acids and Lewis bases. As these interactions are so important, the Lewis acids and bases are given special names (nucleophiles and electrophiles), which are the focus of the next tutorial. For now, notice how curved arrows can be conceptually related to Lewis acids and bases. Curved arrows show that the electrons at the start of the arrow (Lewis base) are being “donated” to the position at the end of the arrow, which “accepts” them (Lewis acid).
Let’s look at some simple reactions involving the charged carbon, nitrogen, and oxygen species introduced in the previous tutorial. These examples aim to (1) show how the concepts of Lewis and Brønsted acids/bases are related to curved arrows and reaction mechanisms, and (2) introduce some of the ways that these charged species can behave in chemical reactions.
It’s not worth memorizing these examples; it’s more important to use them to get a feeling for the different ways that electrons can move in a mechanism.
Carbon
Carbocations
A carbocation contains a positively charged carbon with three bonds and no lone pairs. Carbocations are important intermediates in a number of reactions covered in intro organic chemistry, including the SN1 and E1 reactions.
One pathway for the formation of carbocations starts from alkyl halides. In the following example, a carbocation is formed following the cleavage of the carbon-bromine σ bond, with the bromine taking the bond electrons as a lone pair:
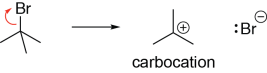
Carbocations can act as Lewis acids, as they can accept a pair of electrons from a Lewis base. In the following example, a lone pair on the oxygen atom of a water molecule (Lewis base) is making σ bond with the carbocation (Lewis acid). Note that the positively charged oxygen is not very favourable, and so it will try to lose a proton (i.e., act as a Brønsted acid).
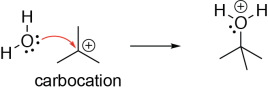
Carbanions
A negatively-charged carbon with three bonds and a lone pair is known as a carbanion. Carbanions are very strong Brønsted bases, as well as Lewis bases. Many of the carbanions you encounter will be stabilized through conjugation with other functional groups (such as carbonyls). Some organometallic molecules (for example, Grignard reagents), which will be introduced later, also behave like carbanions.
In the following example, a carbanion is deprotonating an alcohol. The lone pair on the carbanion carbon forms a σ bond with a hydrogen on the alcohol, breaking the hydrogen-oxygen bond and putting a lone pair on the oxygen. The carbanion here is acting as a Brønsted base (accepting a proton) and a Lewis base (donating an electron pair), and the alcohol is acting as a Brønsted acid (donating a proton) and a Lewis acid (accepting an electron pair).

Nitrogen
Ammonium Ions
An ammonium ion contains a nitrogen with four bonds and no lone pairs. One way that an ammonium ion can be formed is by protonating the corresponding amine. In the following reaction, the lone pair on the amine nitrogen forms a new σ bond with the hydrogen of hydrochloric acid, breaking the hydrogen-chlorine bond, and putting a lone pair on the chlorine. The amine here is acting as a Brønsted base and a Lewis base, and hydrochloric acid is acting as a Brønsted acid and a Lewis acid.
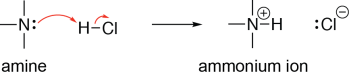
Ammonium ions are acidic (if there is a hydrogen on the nitrogen). In the following example, a hydroxide ion is deprotonating an ammonium ion. A lone pair on the hydroxide oxygen forms a σ bond to the ammonium hydrogen atom, breaking the nitrogen-hydrogen σ bond, and putting a lone pair on the nitrogen. The ammonium ion here is acting as a Brønsted acid and a Lewis acid, and the hydroxide ion is acting as a Brønsted base and a Lewis base:

Amides
An amide contains a negatively-charged nitrogen with two lone pairs and two bonds. However, most of the time, the term ‘amide’ is used to refers to the carbonyl-containing functional group, so it’s worth being aware of this potential ambiguity.
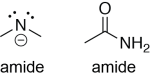
Amides are very strong Brønsted bases. In the following example, the amide ion removes a proton from a ketone. A lone pair on the negatively-charged nitrogen makes a σ bond with a hydrogen on the ketone, breaking the hydrogen-carbon bond, and putting a lone pair on the carbon. The amide ion here is acting as a Brønsted base and a Lewis base, and the ketone is acting as a Brønsted acid and a Lewis acid.

This creates an enolate ion from the ketone, which are important in a number of reactions. While normal carbanions are extremely reactive, having the lone pair on a carbon next to a carbonyl helps to stabilize the charge through conjugation.
Oxygen
Oxonium Ions
An oxonium ion contains a positively-charged oxygen with three bonds (to alkyl groups or hydrogen atoms) and one lone pair. An oxonium ion can be considered a positively-charged form of an alcohol or an ether; there are also oxonium ions based on carbonyl compounds (in which the oxygen is double-bonded to a carbon). The hydronium ion, the simplest oxonium ion, consists of an oxygen with three bonds to hydrogen atoms, and one lone pair (i.e., it is a protonated water molecule, H3O+).
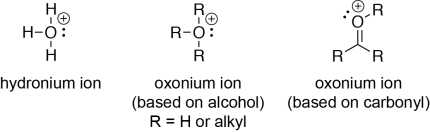
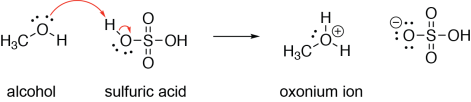
Alcohols and ethers are weak Brønsted bases, but can be protonated when they are treated with a strong Brønsted acid (e.g., sulfuric acid). In the following example, a lone pair on the alcohol oxygen forms a σ bond to a hydrogen atom on sulfuric acid, breaking the sulfuric acid hydrogen-oxygen bond, and putting a lone pair on the oxygen. The alcohol here is acting as a Brønsted base and Lewis base, and sulfuric acid is acting as a Brønsted acid and a Lewis acid.
Oxonium ions are strong Brønsted acids (if there is a hydrogen bonded to the oxygen). In the following example, an oxonium ion protonates an amine, forming an alcohol and an ammonium ion. The lone pair on the amine nitrogen forms a σ bond with a hydrogen atom on the oxonium oxygen, breaking the oxygen-hydrogen bond, and putting a lone pair on the oxygen. The amine here is acting as a Brønsted base and a Lewis base, and the oxonium ion is acting as a Brønsted acid and a Lewis acid.

Alkoxides
An alkoxide contains a negatively-charged oxygen with a bond to an alkyl group, and three lone pairs. Alkoxides are related to the hydroxide ion, which consists of a negatively charged oxygen with three lone pairs and a bond to a hydrogen.
In the following example, an alkoxide is converting an alkyl halide into an alkene, in what is known as an elimination reaction. However, this starts with a typical Brønsted acid-base reaction. A lone pair from the alkoxide oxygen forms a σ bond to a hydrogen on the alkyl halide, and cleaves the hydrogen-carbon bond. The electrons from this bond make a carbon-carbon π bond at the adjacent position, break the carbon-bromine bond, and put a lone pair on the bromine. The alkoxide here is acting as a Brønsted base and a Lewis base, and the alkyl halide is acting as a Brønsted acid and a Lewis acid.
