The steps for naming alkenes build on the steps used for the nomenclature of branched alkanes:
- Identify the parent chain
- Number the parent chain
- List the substituents in alphabetical order
- Finish with the name of the parent chain
Let’s follow these steps to name this alkene:

There are two different sets of rules for identifying the parent chain of an alkene. Recent changes to the IUPAC nomenclature rules say that the parent chain should be the longest chain of carbons, even if this chain does not contain the alkene double bond. However, it is very common to define the parent chain as the longest chain of carbons that includes both carbons in the double bond, so not necessarily the longest chain of carbons. Check which system your instructor expects.
For this alkene, the longest chain is eight carbons long. However, three different eight-carbon chains are possible:

In the first two structures, the double bond is part of the highlighted chain. Furthermore, both highlighted chains have the same substituents at the same positions – both are acceptable as parent chains. The highlighted carbons in the third structure do not include the double bond carbons, therefore this cannot be the parent chain.
Next, we need to number the carbons of the parent chain. For alkenes, the parent chain is numbered so that the double bond carbons have the lowest numbers possible (if the double bond is part of the parent chain – see above). So, for this alkene, numbering starts from the right side, so that the double bond is between carbons 2 and 3.

If the double bond was exactly in the middle of the parent chain, we would number the parent chain to give lower numbers to carbons with substituents.
Next, we name the substituents on the parent chain. There are methyl groups on carbons 2 and 5 (2,5-dimethyl), and a propyl group on carbon 4 (4-propyl). Listing these in alphabetical order (remember that prefixes indicating numbers, like di- and tri-, are ignored), we get: 2,5-dimethyl-4-propyl.
The suffix –ene is used for the name of an alkene parent chain, in place of the –ane suffix used for alkanes. As the parent chain for this alkene is eight carbons long, it is an octene.
We also have to indicate the position of the double bond. This is done by including the number of the first carbon in the double bond (the lowest numbered carbon) in the parent chain name. The IUPAC nomenclature rules say that this number should be in the middle of the alkene name before the -ene suffix, so the parent chain name is oct-2-ene. However, it’s also common to have the number at the beginning of the alkene name (e.g., 2-octene). Check which format is used by your instructor.
Finally, we combine the names of the substituents (in alphabetical order) and the name of the parent chain, giving 2,5-dimethyl-4-propyloct-2-ene:

Next, let’s assign the name of a more complicated alkene:

First, identify the parent chain. There are several different seven-carbon chains for this structure. Two of these chains include both double bonds, and are both acceptable parent chains as they have the same substituents at the same positions:
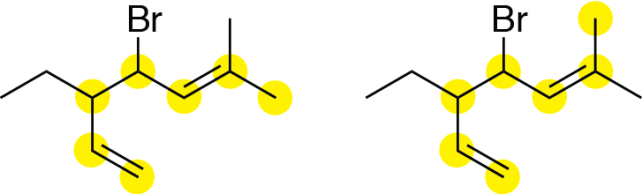
Next, number the parent chain. We have to decide which end of the parent chain to start numbering. As with simpler alkenes, we want to give the lowest numbers possible to the double bond carbons. If we number from the left, the double bonds start on carbons 1 and 5, whereas if we start on the right, the double bonds would start on carbons 2 and 6:

The parent chain has three substituents: an ethyl group on carbon 3 (3-ethyl), a bromine on carbon 4 (4-bromo), and a methyl group on carbon 6 (6-methyl). In alphabetical order, these are: 4-bromo-3-ethyl-6-methyl.
Our alkene parent chain is seven carbons long, corresponding to the alkane heptane. As the parent chain has two double bonds, we use the suffix –adiene in place of the alkene –ene suffix). Thus, the parent chain of this molecule is a heptadiene. If there were three double bonds, we would use –atriene, and so on. We must also indicate the positions of the double bonds using the lower numbered carbon of each double bond, giving us: hepta-1,5-diene.
Combining the substituent names with the parent chain name, we get: 4-bromo-3-ethyl-6-methylhepta-1,5-diene.

Related Topics
Depending on how the substituents on the double-bond of an alkene are oriented, different stereoisomers are possible. The names given to alkenes must distinguish between these stereoisomers using cis-trans notation or E–Z notation.
This tutorial covered the nomenclature of alkenes in which the double bond is part of the parent chain. However, the nomenclature is different if an alkene is a substituent.
The nomenclature of alkenes is also different if there are other functional groups in the molecule (such as alkynes).
Summary
The steps for the nomenclature of alkenes are:
- Identify the parent chain (which depends on the naming rules followed).
- Number the parent chain (giving the lowest possible numbers to the double bond carbons).
- List the substituents in alphabetical order.
- Finish with the name of the parent chain, using the suffix –ene. If the molecule has multiple double bonds, use the suffixes –adiene, -atriene, etc. Indicate the position of the double bond with the number of the lower numbered carbon in the double bond (e.g., but-2-ene).