Benzene is cyclic molecule made up of six carbons, and is drawn with alternating single and double bonds:
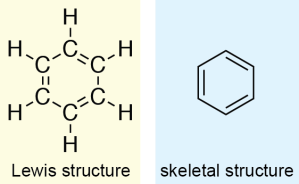
However, these structures are not entirely accurate. The double bonds in the ring are all conjugated (meaning that they are each separated by only one single bond). For benzene, this means that there is another resonance structure that can be drawn; that is, there is another way we can draw the structure which differs only based on where the electrons are located. In this resonance structure, the single bonds are replaced with double bonds, and the double bonds are replaced with single bonds. As resonance structures are different ways of representing the same chemical structure, these two structures represent the same molecule:

The middle double-headed arrow indicates that these two structures are resonance structures.
Average structure…