Background
This tutorial covers the nomenclature of alkyl halides, which are alkanes (saturated hydrocarbons) with one or more halogen atom substituents. The halogens are the elements in group 7 in the periodic table, including fluorine, chlorine, bromine, and iodine.
There are several important alkyl halides which are often called by their common names. It’s worth learning these names:
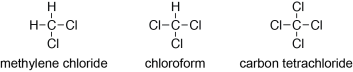
Nomenclature
The nomenclature of alkyl halides is very similar to the nomenclature of branched alkanes: halogen atom substituents are treated the same as alkyl substituents on the parent chain.
The first step is to identify the parent chain, that is, the longest chain of carbons in the molecule. Note that the parent chain does not necessarily have to include the carbons bonded to the halogen atoms.
Next, the carbons of the parent chain are numbered. As with branched alkanes, you want to number the parent chain so that the carbons with substituents have the smallest numbers. Note that alkyl groups and halogen substituents are given the same priority. If you have a situation where you have to decide whether to give an alkyl group or a halogen atom a lower number (see the example below), number the parent chain so that the substituent with alphabetical priority has the lower number.
With the parent chain numbered, you now list the substituents in alphabetical order. To describe a halogen atom as a substituent, replace the suffix –ine with the suffix –o (e.g., from bromine to bromo). Here are the names of the main halogen substituents:

The prefixes di-, tri-, tetra-, and so on are used if there are two or more of a particular halogen atom (e.g., dichloro). Remember that these prefixes are ignored when you are alphabetizing the substituents.
The name given to the parent chain of an alkyl halide is the same as the parent chain name of the corresponding alkane; no special suffixes are used for alkyl halides. The final name is assembled by placing the parent chain name after the list of substituents.
Example

First, identify the parent chain. For this molecule, the longest chain of carbons is six carbons long, highlighted in yellow:
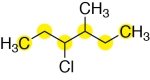
Next, assign numbers to the carbons of the parent chain. If we start numbering from the left, we have 3-chloro and 4-methyl substituents. However, if we start numbering from the right, we have 3-methyl and 4-chloro substituents. As mentioned above, halogen atom substituents and alkyl substituents have the same priority. Therefore, for the tie-breaker, we base the numbering on the alphabetical order of the substituents. The chloro substituent has higher priority, alphabetically, than the methyl substituent – so, we number the parent chain so that the chloro substituent has the lower number.
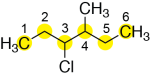
Now, to assemble the full name, we list the substituents (3-chloro and 4-methyl) in alphabetical order. Separate different substituents with hyphens (3-chloro-4-methyl). Finish with the name of the parent chain (with no hyphen between the name of the last substituent and the parent chain). In this example, the six-carbon parent chain is hexane, so the final name is: 3-chloro-4-methylhexane.

Summary
The steps for naming alkyl halides are:
- Identify the parent chain (the longest continuous chain of carbon atoms).
- Number the carbons in the parent chain, such that the carbons with substituents have the lowest numbers possible. Halogen atom substituents are given the same priority as alkyl substituents.
- List the substituents in alphabetical order (ignoring the prefixes di-, tri-, etc for alphabetization).
- Finish with the name of the parent chain (which is not given a special suffix for alkyl halides).